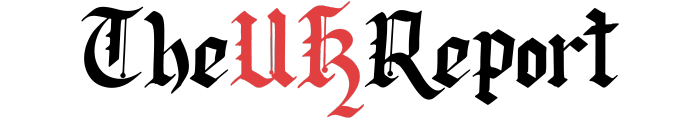In response to an editorial from *The Guardian* on November 23, 2023, regarding the use of animals in medical research, prominent scientists have stressed the necessity of continuing animal testing. They argue that while advancements in alternative methods are important, the complete replacement of animal testing is not yet feasible, particularly in fields requiring complex biological insights.
Dr. Robin Lovell-Badge, a principal group leader at the Francis Crick Institute and president of the Institute of Animal Technologists, acknowledges the value of developing alternatives to animal testing. However, he emphasizes that current strategies primarily focus on regulatory testing, which may not address the challenges of discovery science. He outlined that all researchers in the UK adhere to the 3Rs principle: replacement, reduction, and refinement of animal use. Despite these efforts, he insists that abandoning animal research prematurely could hinder significant scientific discoveries that are crucial for human and animal health.
The discussion highlights significant limitations in existing alternatives. While new approach methodologies (NAMs) have shown promise, they often rely on animal-derived products. For example, organoids and organs-on-a-chip technologies depend on materials such as matrigel, derived from mouse tumors, and foetal bovine serum, which contain vital growth factors that synthetic alternatives cannot yet replicate. This reliance underscores the ongoing need for animal testing in various research contexts.
Prof. Emma Robinson, a professor of psychopharmacology at the University of Bristol, supports Lovell-Badge’s views, stressing that complex biological questions—including those related to disease development and environmental interactions—cannot yet be fully addressed without the use of animal models. She warns that the notion of rapidly replacing animal testing overlooks the complexities of biology and the current limitations of NAMs.
Both scientists advocate for increased investment in NAMs to expedite the development of alternatives that may eventually lead to animal-free research methods. They suggest that additional funding could enhance the validation of these methodologies, particularly in toxicity and pharmacokinetics testing, where current regulations require animal use.
The debate over animal testing continues to evoke strong opinions, reflecting the balance between ethical considerations and scientific necessity. As the dialogue evolves, maintaining rigorous standards for animal welfare remains paramount in the research community. Experts agree that while the future may hold promise for alternatives, the immediate focus should be on ensuring the quality and safety of medical advances through regulated animal research.
In conclusion, as advancements in science strive to reduce reliance on animal testing, the consensus among leading researchers is clear: the complete elimination of animal models in medical research is not yet a viable option. The complexities of biological systems demand a cautious approach, recognizing that while alternatives are being developed, they have not yet reached a stage where they can fully replace animal testing.