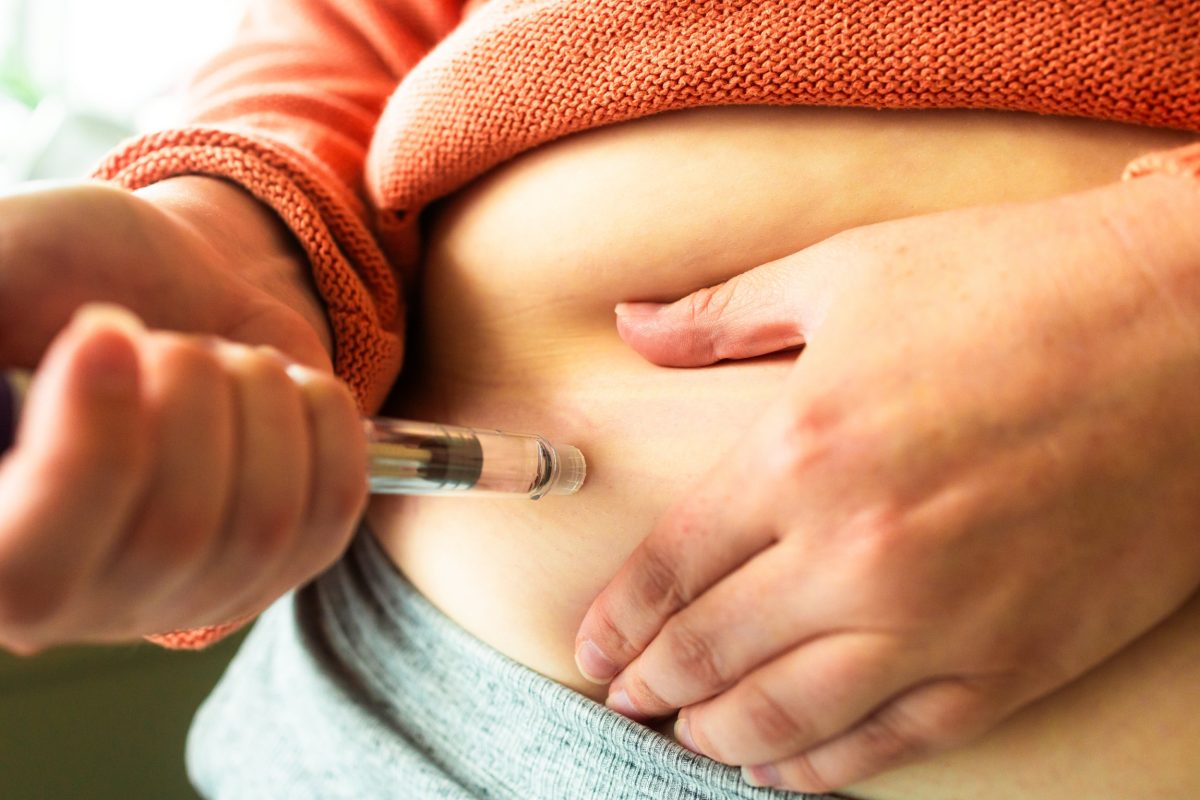
The unapproved weight loss drug retatrutide is drawing attention for its potential effectiveness and significant health risks. Currently available online, this potent injection is being marketed as a revolutionary treatment, but experts warn it poses serious dangers.
Retatrutide, sometimes referred to as the “Godzilla of weight loss injections,” operates similarly to licensed medications like Wegovy and Mounjaro, both of which mimic gut hormones to suppress appetite. Early-stage trials suggest that retatrutide may lead to even greater weight loss; however, these results are yet to be confirmed in larger studies necessary for regulatory approval.
Understanding the Differences
What sets retatrutide apart from its approved counterparts is its enhanced potency. Wegovy, the brand name for semaglutide, typically results in an average weight loss of around 15 percent after one year, while Mounjaro, featuring tirzepatide, can cause up to 21 percent weight loss. In contrast, retatrutide has demonstrated potential for up to 24 percent weight loss within the same timeframe, according to preliminary Phase 2 trials. Notably, participants in these trials continued to lose weight beyond the one-year mark.
Retatrutide’s mechanism involves mimicking three hormones: GLP-1, GIP, and glucagon, earning it the nickname “triple G.” Despite its promising results, it remains unapproved for human use in any country, with Eli Lilly holding the patents for the compound.
Health Risks and Regulatory Concerns
While medically approved injections like Wegovy and Mounjaro are considered relatively safe under physician supervision, the risks associated with unregulated use of retatrutide are substantial. Layla Moran, a Member of Parliament and chair of the Health and Social Care Select Committee, has expressed concern about the public health risks posed by unapproved drugs. She has called on the UK Medicines and Healthcare Products Regulatory Agency (MHRA) to take action against the illegal trade of weight-loss injections.
Moran highlighted that using unlicensed, untested drugs can lead to serious side effects, including pancreatitis and gallbladder disorders. While approved medications require a gradual dosage increase to minimize side effects, those purchasing retatrutide online face higher risks due to the lack of regulation and quality control. Kiran Jones, a pharmacist at Oxford Online Pharmacy, emphasized that individuals might be jeopardizing their health without knowing the exact contents of these products.
Though retatrutide can be found for sale online, it cannot legally be marketed for human use without Eli Lilly’s consent. The chemical formula is publicly available, allowing small manufacturers, particularly in China, to produce and sell it under the guise of “research purposes only.” The product is often sold as powders or ready-to-inject vials, and its popularity seems to have surged among those seeking weight loss or muscle gain.
Eli Lilly has stated that anyone selling retatrutide for human use is acting illegally, and the company has taken action against some online sellers. However, platforms like TikTok and Meta have struggled to fully eliminate advertisements for the drug, making it easily accessible to consumers.
Looking ahead, the final Phase 3 trials for retatrutide are expected to conclude in May 2024. If the results indicate safety and efficacy, it could potentially receive approval in the UK by 2027. Until then, the risks associated with unapproved drugs remain a pressing public health concern. The MHRA has been approached for further comment regarding the ongoing issues surrounding retatrutide and its availability.